The text for this page is original to the Department of Energy's Office of History and Heritage Resources. To learn more about the atom, choose a web page from the menu below: To continue with a quick overview of the Science of the Manhattan Project, jump ahead to the description of Nuclear Physics. Bohr's very influential model proved to be very useful in the description of known experimental results and earned him the Nobel Prize in Bohr postulated that the atomĬonsisted of a hard nucleus, surrounded by electrons orbiting in discrete energy states. In this work Bohr combined Rutherford's atomic model of a massive nucleus with a quantum mechanical theory of energy. Niels Bohr published his hugely influential 1913 paper "On theĬonstitution of Atoms and Molecules," in three parts in Philosophical Magazine. Importantly, Rutherford's model would be picked up and reworked with newly developed quantum mechanical tools by Niels Bohr in what would come to be called the Bohr Atom.
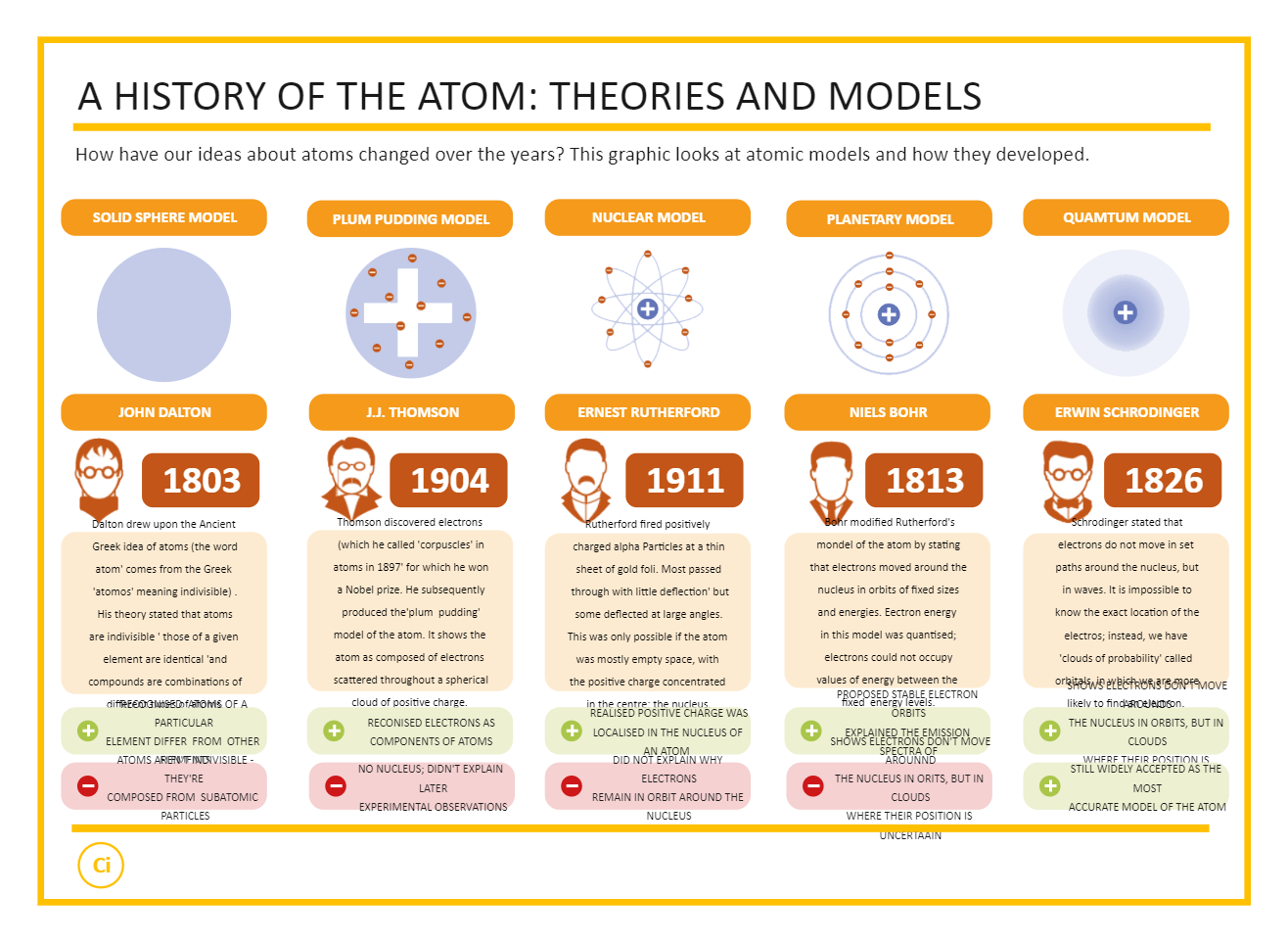
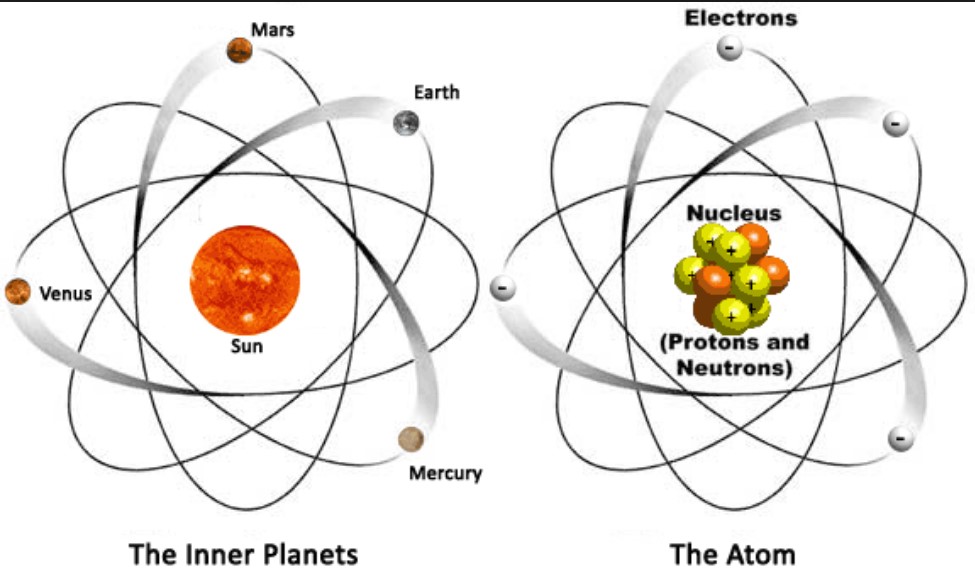
Of a hard, heavy, and electrically charged nucleus surrounded by oppositely charged particles. Through scattering experiments Rutherford argued that an atom was composed With the 1911 publication of Ernest Rutherford's model, ideas about atomic structure began to meaningfully resemble the model accepted today. This approach imagined a positively charged nucleus circled by rings of electrons, but it too suffered from theoretical Same journal as Thomson's model, was Japanese physicist Hantaro Nagaoka's "Saturnian" model of atomic structure. Also published in 1904, the same year and in the Thomson imagined an atomĬomposed of only electrons, and while his model had some promising features, it was not clear to observers how it could be used to describe certain known experimental phenomena. Thomson began developing an early and influential model for atomic structure. With the discovery of the electron in 1897, J.J. To satisfactorily model the structure of atoms, scientists needed to ensure that their models wouldīe stable and that they would be able to account for experimental results. Seaborg).įor many physicists and chemists, the study of atomic structure was an exciting area of research by the turn of the 20th century. Meitnerium (after Lise Meitner) and seaborgium (after Glenn T. Including berkelium (named for the University of California, Berkeley) bohrium (named for Niels Bohr) einsteinium (named for AlbertĮinstein) fermium (after Enrico Fermi) lawrencium (after Ernest O. Scientists have produced at least twenty-three elements in the laboratory, Plutonium, which has ninety-four electrons, is one such element. Yet, scientists can produce even heavier elements in the laboratory with the use of nuclear reactors. Only ninety-two elements occur in nature, from the lightest, hydrogen, having one proton, to the heaviest, uranium, with ninety-two protons. For example, all atoms of uranium have 92 protons, but one type of uranium, the isotope uranium-235 has 143 neutrons This means that atoms of the same element must have the same number of protons, but may have different numbers of neutrons, and consequently differentĪtomic weights. The atomic number, which definesĪ given element, consists solely of the number of protons in the nucleus. Scientists define the atomic mass to be the total number of protons and neutrons. The nucleus is so small compared with the rest of the atom, almost all of the mass of the atom is located there. Protons and neutrons have similar masses, and are about 1,800 times heavier than electrons. This means that if the nucleus was the same size as Earth, the radius of the atom would stretch all the way to the sun. The nucleus is only about 10-12 cm across, while the atom itself is about 10-8 cm. The nucleus is made up of protons and neutrons and makes a tiny fraction of the overall size of the atom. Current models of the structure of the atom consist of a small and relatively heavy nucleus Yet scientists realized that these atoms are themselves are made up of even smaller particlesĪnd at an even smaller scale, scientists now know that neutrons and protons are composed of tiny fundamental particles called quarks.
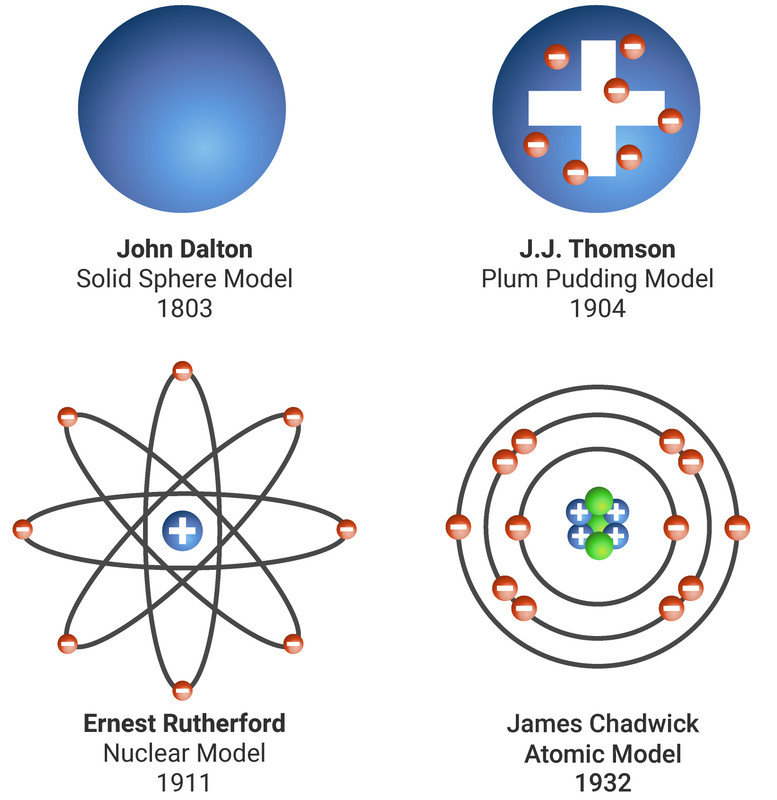
For this reason, they are often considered to be the building blocks of nature. The smallest possible unit of any given element. An atom is a piece of matter that cannot be broken down into smaller units by chemical means, giving
The early twentieth century, when the existence of atoms became widely accepted thanks to the work of Albert Einstein. The composition of these elements was a matter of speculation until Manhattan Project: Science > The Atom and Atomic Structureīy the nineteenth century chemists had recognized that the wide variety of materials that they studied were mixtures of basic, elementary substances.
